
Caramels comprise a group of low anionic charge compounds resulting sucrose thermal decomposition, composed by HMF –hydroxymethylfurfural polymers. HMT does not present visible colour, but when polymerized it forms high intense colour compounds.
The reaction of caramels formation is much slower than those of melanoidins and HADP formation (Mersad et al., 2000). By this fact these colourants have not great contribution in colour of juices and syrups (Paton and McCowage, 1987).
For caramels formation, sugar temperature must be higher than its melting (185 ºC) (Ahari and Genetolle, 1961). Although process temperatures are in average much lower than this, it can happen that sugar be accidentally in contact with surfaces at temperatures higher enough to produce caramelization. Technical faults as steam valves leakage, entrance of sugar solutions in steam pipes, can happen, with consequent caramel formation.
In the next figure it is presented the spectrum of caramel solution at pH 9 (Bento 1995a). Spectra at pH 11 and pH 3 present peaks at 229nm and 285nm (Bento, 2002).

Spectrum of Caramels at pH 9
In the next figure it is presented the diagram obtained by GPC with ELS detector using a caramel solution(Bento and Sá, 1998). Caramels present one peak at 51,9 minutes of retention time, corresponding to a molecular weight of 5,5kD.
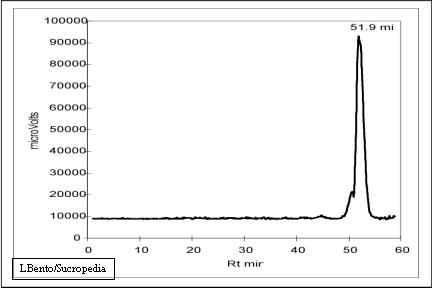
ELS diagram of Caramels
In the next figure it is presented the UIV curve of caramels (Bento, 2002). UIV is obtained by the quotient of attenuancie sat pH 9 and pH 3 in the UV spectrum. As it is observed this curve do not present a great variation between present 240nm e 300nm.
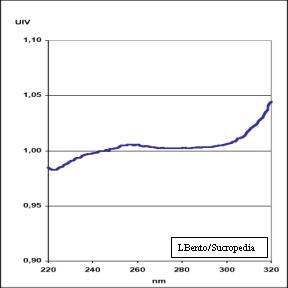
UIV curve of Caramels
In the next figure it is presented the caramels chromatogram obtained by sucrose chromatography (Bento, 1994). In this chromatogram it is observed that these colourants present a single peak eluted at 19 minutes of Rt, with 1-propanol as eluent. This result shows that caramels present some degree of hydrophobicity.
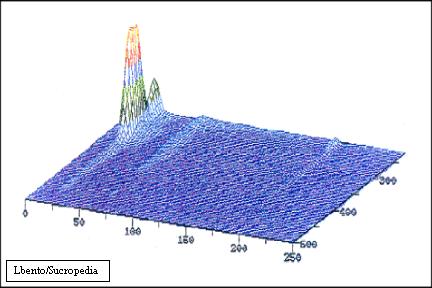
Sucrose chromatogram of Caramels
Bibliography
Ahari D., J. Genotelle, 1961, La coloration en sucrerie: perfectionements apportes a sa mesure, Ind. Alim.
Agric., 742-747
Bento L.S.M., 1995, Application of UV Spectrophotometry to study sugar colourants throughout the refining
process, Proc. of S.I.T. Conf., 211-230
Bento L.S.M., 2002, Separation of beet and cane colourants through styrenic strong base resins, Proc. of
S.P.R.I. Conf., 311-327
Bento L.S.M., S. Sa, 1998, sudy of HMW compounds in sugar liquors from carbonatation and ion-exchange
resins using GPC chromatography with an evaporative light scattering detector, Proc. of S.P.R.I.
Conf., 233-247
Mersad A., R. Lewandowski, M. Decloux, 2000, Colorants in cane sugar industry, Proc. of S.I.T. Conf., 235-
247
Paton N.H., Mc Cowage, 1987, Colour forming mechanisms during mill processing, Proc. of Aust. S.S.C.T.
Conf., 11-20
E0048/58